ISSN: 1659-455X • e-ISSN: 1659-407X
Vol. 18 (1), enero-junio 2026
Recepción 28 mayo 2025 • Corregido 4 noviembre 2025 • Aceptado 4 noviembre 2025
DOI: https://dx.doi.org/10.15359/revmar.18-1.1
A new fiddler crab species (Brachyura: Ocypodidae) from the Pacific coast of Panama
Una nueva especie de cangrejo violinista (Brachyura: Ocypodidae) del Pacífico de Panamá
Roberto C. Lombardo González1,2,3*
ABSTRACT
A new fiddler crab species of the genus Leptuca, discovered at five sites along the Panamanian Pacific coast, is described here. Specimens were collected from mangrove edges during diurnal low tides between June 2024 and August 2025. Morphological traits were consistent across all sites, with an average carapace width of 12.53 ± 1.81 mm. Diagnostic features include bacilliform, V-shaped patches of pubescence on the mesogastric region; a squared manus carpal cavity with a distinct distal beaded edge; a robust oblique ridge elevated into a triangular projection; a dactyl ventral margin with a large, rounded tooth; and paired beaded ridges on the posterior surface of the second or third ambulatory carpi. The proposed species name is Leptuca bacillopilosa sp. nov., in reference to the characteristic symmetrical rod-like pile patches on the carapace. This discovery enhances the understanding of fiddler crab diversity in the region and highlights the significance of continued taxonomic surveys.
Keywords: oblique ridge, carapace, Leptuca, pubescence, tubercle.
RESUMEN
Se describe una nueva especie de cangrejo violinista del género Leptuca proveniente de cinco sitios a lo largo de la costa pacífica panameña. Los especímenes fueron colectados entre junio de 2024 y agosto de 2025 en los márgenes de los manglares durante mareas bajas diurnas. Los rasgos morfológicos fueron consistentes entre sitios, con un ancho promedio de caparazón de 12.53 ± 1.81 mm. Los caracteres diagnósticos incluyen parches de pubescencia baciliformes en forma de V en la región mesogástrica; cavidad carpal del manus cuadrada con un margen distal fuertemente tuberculado; cresta oblicua robusta y elevada en una proyección triangular; margen ventral del dáctilo con diente redondeado grande; y par de bordes granulados en la superficie posterior del segundo o tercer carpo ambulatorio. Proponemos el nombre Leptuca bacillopilosa sp. nov. en referencia a los parches diagnósticos simétricos de pubescencia en forma de bacilo en el caparazón. Este hallazgo contribuye a la diversidad taxonómica de los cangrejos violinistas en la región y subraya la importancia de la continuidad de los inventarios taxonómicos.
Palabras clave: borde oblicuo, caparazón, Leptuca, pubescencia, tubérculo
Since Crane’s 1975 monograph Fiddler Crabs of the World, which recognized 62 species, the number of described species has grown, reaching 107 in the most recent compilation on the Fiddler Crabs website (Rosenberg, 2014a). In Panama, seven species occur on the Caribbean coast, distributed among three genera: four species of Minuca, two of Uca, and Leptuca thayeri (Lombardo, 2025). On the Pacific coast, recent studies have added three new country records: Minuca osa Landstorfer & Schubart, 2010 (Lombardo, 2022), Minuca zacae Crane, 1941 (Lombardo, 2024a) and Leptuca tallanica von Hagen, 1968 (Lombardo, 2024b). With these additions, the number of Pacific species rises to 31, representing four genera: Leptuca (17 species), Uca (6 species), Minuca (7 species), and Petruca panamensis (Rosenberg, 2014b; Shih et al. 2015; Rosenberg, 2020). In total, 38 species are known from both coasts (Table 1).
Table 1. Checklist of fiddler crab species recorded in Panama, organized by coast (Caribbean and Pacific), with taxonomic authority. Presence was determined based on Lombardo (2025); geographic ranges were documented on the Fiddler Crabs website; and records were verified in the citizen science platform iNaturalist
Cuadro 1. Lista de especies de cangrejos violinistas registradas en Panamá, organizadas por costa (Caribe y Pacífico), con la autoridad taxonómica. La presencia se determinó a partir de Lombardo (2025), rangos de distribución documentados en el sitio web Fiddler Crabs y registros verificados de la plataforma de ciencia ciudadana iNaturalist
|
Coast |
Species |
Authority |
|---|---|---|
|
Caribbean |
Leptuca thayeri |
Rathbun, 1900 |
|
Minuca burgersi |
Holthuis, 1967 |
|
|
Minuca mordax |
Smith, 1870 |
|
|
Minuca rapax |
Smith, 1870 |
|
|
Minuca vocator |
Herbst, 1804 |
|
|
Uca maracoani |
Latreille, 1802 |
|
|
Uca major |
Herbst, 1796 |
|
|
Pacific |
Leptuca batuenta |
Crane, 1941 |
|
Leptuca beebei |
Crane, 1941 |
|
|
Leptuca deichmanni |
Crane, 1941 |
|
|
Leptuca dorotheae |
Crane, 1975 |
|
|
Leptuca festae |
Nobili, 1901 |
|
|
Leptuca inaequalis |
Crane, 1941 |
|
|
Leptuca latimanus |
Rathbun, 1898 |
|
|
Leptuca limicola |
Crane, 1941 |
|
|
Leptuca oerstedi |
Milne-Edwards, 1852 |
|
|
Leptuca pygmaea |
Crane, 1941 |
|
|
Leptuca saltitanta |
Crane, 1941 |
|
|
Leptuca stenodactylus |
Milne-Edwards, 1873 |
|
|
Leptuca tallanica |
von Hagen, 1968 |
|
|
Leptuca tenuipedis |
Crane, 1941 |
|
|
Leptuca terpsichores |
Crane, 1941 |
|
|
Leptuca tomentosa |
Crane, 1941 |
|
|
Leptuca umbratila |
Crane, 1941 |
|
|
Minuca argillicola |
Crane, 1941 |
|
|
Minuca brevifrons |
Stimpson, 1860 |
|
|
Minuca ecuadoriensis |
von Hagen, 1987 |
|
|
Minuca galapagensis |
Maccagno, 1928 |
|
|
Minuca herradurensis |
Crane, 1941 |
|
|
Minuca zacae |
Crane, 1941 |
|
|
Minuca osa |
Landstorfer & Schubart, 2010 |
|
|
Petruca panamensis |
Rathbun, 1914 |
|
|
Uca heteropleura |
Smith, 1870 |
|
|
Uca insignis |
Milne-Edwards, 1873 |
|
|
Uca intermedia |
Crane, 1941 |
|
|
Uca ornata |
Smith, 1870 |
|
|
Uca princeps |
Smith, 1870 |
|
|
Uca stylifera |
Milne-Edwards, 1873 |
Despite these additions, recent sampling along Panama’s Pacific coast has uncovered specimens that seem not to match known species descriptions (Fig. 1). Preliminary morphological examination indicates that these specimens can be placed within the Leptuca genus, as they feature a frontal region 25 - 30% of the carapace width, laterally semi-cylindrical carapace profile, and short anterolateral margins sloping posteriorly. These characters distinguish them from other genera: Uca (narrow fronts), Minuca (long convex margins), and Petruca (angular, bead-edged carapaces) (Shih et al. 2015). In addition, the major chelipeds show reduced ornamentation and a distinct ridge pattern typical of Leptuca, differing from the irregularly tuberculate chelipeds of Minuca (Bott, 1973; Crane, 1975; Shih et al. 2016).

Fig. 1. Leptuca bacillopilosa sp. nov. images from the Panama Pacific. Male specimens are shown in panels A-B (Horconcitos), and female C (Horconcitos) and D (El Agallito); see link for Appendix 1 video
Fig. 1. Imágenes de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Ejemplares machos en los paneles A-B (Horconcitos), y hembras en C (Horconcitos) y D (El Agallito); ver vínculo para video del apéndice 1
The new country records underscore gaps in our understanding of fiddler crab diversity (Lombardo, 2024b) and support the hypothesis that these morphologically distinct specimens may represent a previously undescribed species. Therefore, detailed morphological analyses are needed to clarify the taxonomic identity of these specimens. The discovery of a new species is particularly significant given the ecological importance of fiddler crabs in intertidal mudflats and mangroves, where they contribute to sediment turnover, nutrient cycling, and substrate aeration through their burrowing activity (Mokhtari et al. 2015; Booth et al. 2019). They also serve as prey for a variety of coastal predators and are important bioindicators of environmental health (Lombardo, 2023; Xu et al. 2024). Thus, the objective of the present study was to clarify the taxonomic identity of the observed specimens from five sites along the Pacific coast of Panama through detailed morphological description and comparison with known species.
Fiddler crabs were collected from five sites in the Panamanian Pacific between June 2024 and August 2025 (Fig. 2). Collections were made at El Agallito, Herrera province (8° 0ʼ 8.12ʼʼ N, 80° 24ʼ 10.37ʼʼ W) in June 2024; Horconcitos, Chiriquí Province (8° 19ʼ 18.66ʼʼ N, 82° 9ʼ 31.87ʼʼ W) in November 2024; Lagartero, Veraguas (7° 39ʼ 45.83ʼʼ N, 81° 12ʼ 21.75ʼʼ W) in December 2024; Búcaro, Los Santos province (7° 21ʼ 19.01ʼʼ N, 80° 22ʼ 13.07ʼʼ W) in January 2025; and Puerto Yurre, Chiriquí province (8° 6ʼ 33.77ʼʼ N, 81° 41ʼ 52.06ʼʼ W) in August 2025.

Fig. 2. Relative location of sampling sites from El Agallito (AG), Horconcitos (HO), Lagartero (LG), Búcaro (BU), and Puerto Yurre (YU), Pacific coast of Panama. Scale = 50 km
Fig. 2. Ubicación relativa de los sitios de muestreo en El Agallito (AG), Horconcitos (HO), Lagartero (LG), Búcaro (BU) y Puerto Yurre (YU), Pacífico panameño. Escala = 50 km
Specimens were observed at the edges of mangroves during diurnal low tides, over soft, muddy substrates, using binoculars (Bushnell 10×50) and a camera (Sony Alpha 5 100 with a Tamron 50 - 400 mm f/4.5 - 6.3 lens). Eight males and two females were collected at each site for morphological analysis (N = 50). In the laboratory, crabs were washed and then euthanized in a freezer (10 min). Specimens were analyzed using a stereoscope (SZ2-ILST) and microscope (CX43), equipped with a camera (EP50) and EPview software (Olympus, Japan). Carapace width (CW), carapace length (CL), frontal width (FW), chela height (ChH; vertical dimension of the propodus), chela length (ChL; length along the propodus longest horizontal axis), as well as dactyl length (DL), were measured using a Vernier caliper (0.01 mm). The size frequency distribution was determined for males and females. Morphometric relationships were analyzed using regression based on pooled data, to examine the relationships between CW and CL, and between FW and CW as well as the FW-CW ratio. For males, ChH, ChL, DL and DL to ChL ratio were regressed against CW to evaluate proportional changes in claw morphology relative to size. Species description was conducted following literature (Crane, 1941; von Hagen, 1968; Crane 1975; Rosenberg, 2001; Beinlich & von Hagen, 2006; Shih et al. 2016; Rosenberg, 2019; Shih & Chan, 2022; Thurman et al. 2025). Reference specimens were deposited at the G. B. Fairchild Museum of Invertebrates at Universidad de Panamá, under access code MIUP-H0087.
Taxonomy
Family Ocypodidae Rafinesque, 1815
Genus Leptuca Bott, 1973
Leptuca bacillopilosa sp. nov.
ZooBank registration
urn:lsid.org:act:6AF5470D-29BD-4D3C-A4F3-27D9516676B7.
Holotype
Adult male (CW = 13.04 mm; CL = 6.41 mm), deposited in the G. B. Fairchild Museum of Invertebrates, Universidad de Panamá, catalog number MIUP-H0087; Panama, Pacific coast, El Agallito, Herrera Province (8° 0ʼ 8.12ʼʼ N, 80° 24ʼ 10.37ʼʼ W), collected by R. Lombardo, 19 October 2024. Additional material examined includes adult males and females; see Materials and Methods.
Morphometry
The average CW of crabs was 12.53 ± 1.81 mm SD (Table 2; Fig. 3A). The relationship between CL and CW was significant (ANOVA, F(1, 48) = 13.99, P < 0.001; R2 = 0.23; Fig. 3B). Front width increased with carapace width CW (ANOVA, F(1, 38) = 17.93, P < 0.001; R2 = 0.32; Fig. 3C), with an estimated slope of 0.08 (SE = 0.02, t(38) = 4.23, P < 0.001) and a significant intercept of 1.90 (SE = 0.23, t(38) = 8.36, P < 0.001). However, the fronts of larger crabs grow proportionally slower (ANOVA, F(1, 38) = 65.32, P < 0.001; R2 = 0.63; Fig. 3D). The slope for this regression was negative (-0.42 ± 0.05, t(38) = -8.08, P < 0.001). Neither ChL nor DL significantly predicted CW. Stepwise elimination revealed ChH as a significant positive predictor (b = 0.58, SE = 0.19, t(38) = 2.99, P = 0.005; R2 = 0.20; Fig. 3E). When DL was expressed relative to ChL (DL-ChL ratio: 62.1%), the relationship with CW was strong (ANOVA, F(1, 38) = 321.49, P < 0.001; R2 = 0.89; Fig. 3F). The slope was 1.45 (lack-of-fit, P = 0.86; SE = 0.08, t(38) = 17.93, P < 0.001).
Table 2. Leptuca bacillopilosa sp. nov. biometrics from five sites in the Panamanian Pacific. Variables are given in millimeters
Cuadro 2. Biometría de Leptuca bacillopilosa sp. nov. de cinco sitios en el Pacífico panameño. Variables dadas en milímetros
|
Variable |
Sex |
N |
Mean ± SD |
Min. |
Max. |
|
Carapace width |
♂ |
40 |
12.82 ± 1.80 |
9.75 |
18.36 |
|
Carapace length |
7.23 ± 1.55 |
4.49 |
10.54 |
||
|
Major chela length |
16.58 ± 5.28 |
7.35 |
27.97 |
||
|
Major chela height |
6.64 ± 1.35 |
4.03 |
10.00 |
||
|
Carapace width |
♀ |
10 |
11.36 ± 1.38 |
9.31 |
12.87 |
|
Carapace length |
6.71 ± 1.10 |
5.00 |
8.38 |
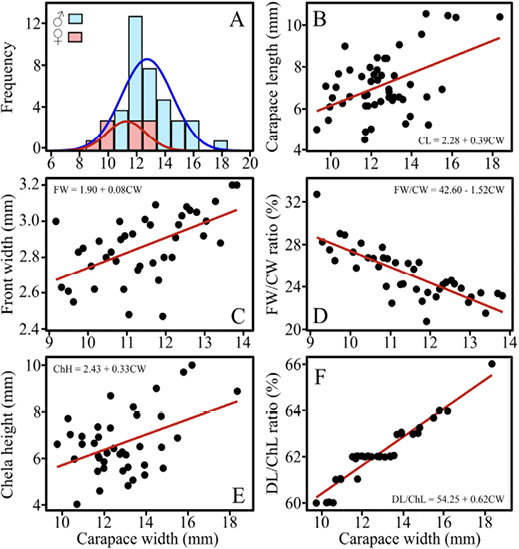
Fig. 3. Morphological features in the fiddler crab Leptuca bacillopilosa sp. nov.
Male and female frequency distribution of carapace width (CW) (A). Relationships shown (B-F): carapace length (CL) with CW (B), front width (FW) with CW (C), the FW/CW ratio with CW (D), chela height (ChH) with CW (E), and the DL/ChL ratio relative to CW (F)
Fig. 3. Características morfológicas en el cangrejo violinista Leptuca bacillopilosa sp. nov. Distribución de frecuencia del ancho del caparazón (CW) para machos y hembras (A). Relaciones mostradas (B-F): longitud del caparazón (CL) con CW (B), ancho frontal (FW) con CW (C), la proporción FW/CW con CW (D), altura de la quela (ChH) con CW (E), y la proporción DL/ChL en relación con CW (F)
Diagnosis
Specimens display diagnostic features of the family Ocypodidae and subfamily Gelasiminae, including a rectangular carapace, wide fronto-orbital region, convex or slightly angular front without a median notch, and elongated eyestalks with corneas less than half the peduncle length (Crane, 1975; Shih et al. 2016). Within the genus Leptuca, the specimens are distinguished by the following combination of characters: (1) bacilliform carapace pubescence forming symmetrical V-shaped patches on the mesogastric region in both sexes, with females additionally exhibiting circular basal pile patches overlying the lateral stria; (2) major cheliped dactylus with mid-row tubercles fusing into a large, round tooth, sometimes with a tricuspid pollex tooth, the dactylar and pollex teeth offset; (3) major manus with a squared carpal cavity and sharply demarcated beaded distal edge with an oblique ridge forming a central triangular projection; (4) outer manus with dorsally larger tubercles arranged in a gradient and a beaded ventral keel with a shallow groove; (5) two posterior beaded ridges diverging anteriorly on the second and/or third ambulatory carpus; and (6) gonopod with underdeveloped flanges, a small thumb ending proximal to the corneous pore base, and a broad, flat inner process nearly reaching the pore.
Etymology
We propose the specific epithet bacillopilosa which derives from the Latin “bacillus” (rod) and “pilosus” (hairy), in reference to the diagnostic bacilliform pubescence forming symmetrical V-shaped patches on the mesogastric region of the carapace (Fig. 4B-C. e). An identification key for L. bacillopilosa and the other sympatric Leptuca species, including the Caribbean congener L. thayeri is contained in Table 3.
Table 3. Morphological identification key for sympatric Leptuca species from five sites in the Panamanian Pacific, including Leptuca bacillopilosa sp. nov. and incorporating the Caribbean congener Leptuca thayeri
Tabla 3. Clave de identificación morfológica para especies de Leptuca simpátricas con Leptuca bacillopilosa sp. nov. en cinco sitios del Pacífico panameño, incorporando el congénere caribeño Leptuca thayeri
|
Item |
Description |
Leads to |
|---|---|---|
|
1a |
Front < 15% of carapace width; eyestalks long and slender; major cheliped pollex and dactyl broad and flat, with dactyl tip never curving over the pollex. |
Uca |
|
1b |
Front ≥ 15% of carapace width; major cheliped variable, with dactyl often curving over the pollex. |
2 |
|
2a |
Front > 31% of carapace width; carapace laterally non-cylindrical; anterolateral margin long, curving into the dorsolateral surface; always two posterolateral striae; pleonites free. |
Minuca |
|
2b |
Carapace faintly striated or pubescent; posterolateral striae: ranging from 0–2; oblique ridge on inner manus variable; carpal cavity may possess a beaded edge; ventral pollex tuberculate ridge variable. |
3 |
|
3a |
Dorsal carapace nearly flat and smooth, with a single posterolateral stria; no ventral tuberculate ridge on pollex; major chela smooth; tips of minor fingers bearing a brush of long setae. |
Petruca panamensis |
|
3b |
Front 19-30% of carapace width; carapace laterally arched to semi-cylindrical; anterolateral margin short; dactylus and pollex generally longer than the manus. |
Leptuca; 4 |
|
4a |
Front 19-30% of carapace width; carapace with numerous, often large, patches of pubescence; orbits oblique; anterolateral margin short; dorsolateral margins strongly convergent; palm oblique ridge variable; smooth to variable beaded posterior surface of the second and/or third ambulatory carpi. |
5 |
|
4b |
Front 19-30% of carapace width; dorsal carapace with minor patches of pubescence; pollex projections variable. |
6 |
|
4c |
Front 20-30% of carapace width; dorsal carapace without pubescence. |
7 |
|
5a |
Western Atlantic realm species, distributed throughout the Caribbean islands. Large crab with a front measuring 22% of carapace width; pollex tip curving down; palm exhibiting a dorsal beaded edge above the carpal cavity, which does not curve down around the distal margin of the cavity. |
L. thayeri |
|
5b |
Large crab with a front measuring 19% of carapace width. Dorsal carapace with profuse pubescence. Inner carpus bears a large tooth. Beaded distal edge of carpal cavity absent, and upper palm is finely granulated. Ambulatory meri are broad with strongly convex dorsal margins. Ambulatories have persistent pile, especially in females and juveniles. Pair of beaded ridges present on the posterior surface of the second and/or third ambulatory carpi. |
L. umbratila |
|
5c |
Front 24% of carapace width. Pubescence concentrated in the H-depression and branchial region; major cheliped has a narrow gape; dactyl prehensile edge bears 3-5 large, rounded teeth proximally; beaded distal edge of carpal cavity is weak or absent and granulated; male ambulatories are glabrous, while pile is present on females; single ridge present on posterior surface of ambulatory carpi. |
L. tomentosa |
|
5d |
Front 25% of carapace width; bacilliform V-shaped pubescence patches on male mesogastric region; females with circular basal pubescence, covering a lateral stria; carpal cavity squared with continuous distal beaded border; oblique ridge elevated forming triangular projection; dactyl ventral margin with three rows fusing into large rounded tooth; tricuspid tooth on pollex; diverging beaded ridge on posterior second and/or third ambulatory carpi surface, most evident on females. |
L. bacillopilosa sp. nov. |
|
5e |
Very small crab (carapace width ≤ 8 mm); major manus is swollen, outer manus and palm are coarsely tuberculate; large tooth present on the proximal dactyl and pollex; first ambulatory lacks anterior tubercles. |
L. pygmaea |
|
6a |
Small crab with a front 26% of carapace width. Carapace pubescence J-shaped posteriorly. Fingers are blade-like, with the dactylus terminus broad. Major pollex bears a distal triangular projection. |
L. batuenta |
|
6b |
Small crab with a front 28% of carapace width. H-depression features eight dorsal pubescence patches arranged in a rectangle. Palm oblique ridge is weak, bearing a row of small distal tubercles. First ambulatory has anterior tubercles on both the merus and carpus. |
L. inaequalis |
|
6c |
Front 20% of carapace width. Anterolateral margins are straight, elongate, markedly divergent, and abruptly angled posteriorly. Carapace branchial region has 2–6 patches of pile. Major chela is short, with fingers longer than the manus. Gape pile is copious. Pollex is triangular and broader proximally than the dactyl, with a triangular projection at mid-length. |
L. oerstedi |
|
6d |
Front 30% of carapace width. Males have six coma-shaped pile patches on the dorsal carapace, while females have two. A large distal tubercle is present at about two-thirds the length of the pollex. First ambulatory displays anterior tubercles on the carpus only. |
L. tallanica |
|
7a |
Dactyl and pollex of the large claw are much shorter than the manus. Tuberculate ridge is reduced or absent. |
8 |
|
7b |
Dactyl and pollex of the large claw are much longer than the manus. |
9 |
|
8a |
Major chela gape is extremely narrow. Fingers are shorter than the manus. Palm is smooth. First ambulatory carpus on the major side features a broad, low crest of granules running longitudinally. |
L. latimanus |
|
8b |
Very small crab. Major dactyl bears two large, triangular projections. Gape is narrow. Pollex is broader than the dactylus at the base, with the end of the pollex varying from strongly scalloped to a pointed terminus. |
L. saltitanta |
|
8c |
Ambulatories are particularly long and slender in both sexes, with straight dorsal margins of the meri. No large projections are present on the dactyl or at the articulation gap. Pollex is triangular and broader than the dactylus, with a single large tubercle located approximately halfway along the dorsal pollex edge. |
L. tenuipedis |
|
9a |
Large cheliped dactyl and pollex are blade-like, with one predactyl ridge. Small chela bears large, unequal serrations. |
10 |
|
9b |
Large cheliped dactyl and pollex are tubular, with two predactyl ridges. Dactyl-pollex is very long, and the gape is wide with the dactyl strongly arched. |
11 |
|
10a |
Carapace is strongly arched, with a longitudinal shallow furrow on the male branchial region. Major chela upper, outer manus displays a linear arrangement of minute granules. Pollex always has one subdistal, tuberculate tooth. Palm lacks a stridulating ridge. |
L. deichmanni |
|
10b |
Minor chela is slender and pointed, not meeting perfectly. Pubescence occurs at the dactyl-pollex junction, and teeth are small. A triangular sulcus is present at the pollex-manus junction. Margins of the middle ambulatory meri are straight in males, while females have pile on the posterior sides of the carapace. |
L. stenodactylus |
|
10c |
Dactyl-pollex is long, with the gape smaller than the width of the dactyl or pollex. Gap is small, and the pollex has a large central tooth. Pubescence is present at the dactyl articulation. Tips of the small chela are obliquely truncate and meet seamlessly. First major ambulatory leg bears anterior tubercles on the carpus and manus. Margins of the middle ambulatory meri are slightly convex in males; females lack pile on the posterior sides of the carapace. |
L. beebei |
|
11a |
Major cheliped fingers are strikingly long, measuring 3–7 times the manus length. Dactylus is longer than the pollex and curves smoothly. Male carapace is dorsally without pile, while females have persistent pile postero-dorsally and posteriorly on the sides. The third and fourth female ambulatories have pile extending from the merus to the propodus. |
L. festae |
|
11b |
Arching of the dactyl over the pollex is strong, with the pollex remaining straight and pubescence present at the dactyl-manus junction. The first ambulatory leg has distal anterior tubercles on the merus, carpus, and manus. Female carapace lacks posterior lateral pile. |
L. dorotheae |
|
11c |
Posterior ventral surface of the manus has 6–7 stridulating ridges. Front measures 29% of carapace width, with the anterior margin of the front obsolescent. |
L. terpsichores |
Description
Carapace. Pentagonal (Fig. 4) and moderately arched in lateral view (Fig. 5A♂-B♀). Front is narrow (FW-CW ratio: 25.2%), but not as in Uca (Fig. 6A-B. a); anterior front edge distinct (Fig. 6B. e). Supraorbital ridges are sinusoidal and clearly marked by a thin edge; their width spans almost the entire orbital cavity, while their height is approximately 47% less than that of the resting eyestalk (Fig. 6D. b, g). Suborbital crenations are present, minute proximally, increasing towards orbital channel. Larger crenations are square-shaped and separated (Fig. 6C-D. f). The antero-lateral angles are almost square (Fig. 4A, C. a), and the antero-lateral margins are short (Fig. 4A, C. b). The dorso-lateral margins slope inward, more markedly in males than in females (Fig. 4A, C. c), and extend down to the lower level of the mesogastric region, where they end straight (Fig. 4B, D. c). A single posterolateral stria is located below the distal portion of the dorsolateral margin (Fig. 4B. d). The vertical-lateral margin is complete, finely beaded, sigmoidal, and curves posteriorly to meet the lower carapace edge between the third and fourth ambulatory coxae (Fig. 5A-B. a). The carapace dorsal surface is smooth, scattered setae are often present. In males, there are two symmetrical, V-shaped patches of bacilliform pubescence on either side of the mesogastric region (Fig. 4B-C. e). In females, similar pubescence is present; each setal patch (pile) has a circular basal area that overlies and partially covers the lateral stria. Each patch originates at the posterior corners of the carapace and extends inward and upward, with elongated and irregularly shaped pile patches ascending the carapace surface toward the midline (Fig. 4D. f).

Fig. 4. Carapace morphological characters of Leptuca bacillopilosa sp. nov. from Panama Pacific. Carapace dorsal and postero-dorsal views (A-B♂; C-D♀). Structures: a, antero-lateral angle; b, antero-lateral margin; c, dorso-lateral margin; d, postero-lateral striae; e, carapace mesogastric region pile (♂); f, posterior carapace pile (♀). Scale = 3 mm
Fig. 4. Caracteres morfológicos del caparazón de Leptuca bacillopilosa sp. nov. del Pacifico de Panamá. Vistas dorsal y postero-dorsal del caparazón (A-B♂; C-D♀). Estructuras: a, ángulo antero-lateral; b, margen antero-lateral; c, margen dorso-lateral; d, estrías postero-laterales; e, pubescencia de la región mesogástrica del caparazón (♂); f, pubescencia posterior del caparazón (♀). Escala = 3 mm

Fig. 5. Carapace lateral view of Leptuca bacillopilosa sp. nov. from the Panamanian Pacific (A♂; B♀). Structure: a, vertical-lateral margin. Scale = 2 mm
Fig. 5. Vista lateral del caparazón Leptuca bacillopilosa sp. nov. del Pacífico panameño (A♂; B♀). Estructura: a, margen vertical-lateral. Escala = 2 mm

Fig. 6. Anterior view of Leptuca bacillopilosa sp. nov. from the Panamanian Pacific. Frontal view (A), front and distal edge detail (B), suborbital ridge (C), upper-orbital edge (D). Structures: a, front; b, eyestalk; c, ocular orbit; d, third maxilliped; e, anterior front edge; f, suborbital crenations; g, sinusoidal edge. Scale: A-B = 1.5 mm; C-D = 0.8 mm
Fig. 6. Vista anterior de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Vista frontal (A), detalle del borde frontal distal (B), borde suborbital (C), borde supra-orbital (D). Estructuras: a, frente; b, pedúnculo ocular; c, órbita ocular; d, tercer maxilípedo; e, borde antero-frontal; f, crenulaciones suborbitales; g, borde sinuoso. Escala: A-B = 1.5 mm; C-D = 0.8 mm
Major cheliped merus-carpus. Merus has an undefined postero-dorsal margin and a blunt antero-dorsal margin (Fig. 7A. a) bearing small distal spines (Fig. 7A-B. b); dorsal surface near the base smooth; ventral margin bluntly angular with a weakly serrated edge (Fig. 7B. c); anterior and postero-ventral surfaces are nearly smooth. Carpus is nearly smooth with pubescence on distal postero-dorsal surface; the pile process continues into the proximal manus outer surface and edge of the carpal cavity. Mesial surface of the carpus is armed with small tooth-like tubercles (4-6) in an oblique row, adjacent to the merus joint (Fig. 7B. d).

Fig. 7. Major cheliped merus of Leptuca bacillopilosa sp. nov. from Panama Pacific. Merus antero-dorsal view (A), merus and carpus anterior surface (B). Structures: a, blunt antero-dorsal margin; b, antero-dorsal distal spines; c, ventral margin serration; d, inner carpus tuberculation. Scale = 3 mm
Fig. 7. Mero del quelípedo mayor de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Vista antero-dorsal del mero (A), superficie anterior del mero y carpo (B). Estructuras: a, margen antero-dorsal romo; b, espinas distales antero-dorsales; c, margen ventral serrado; d, tubérculos carpales internos. Escala = 3 mm
Major manus. The outer upper manus surface is covered with medium to very small tubercles, in a gradient, where the largest are on the upper half (Fig. 8A. a); the lower half is moderately smooth (Fig. 8A. b). The dorsal margin is proximally bent (Fig. 8B-C. c), while the manus is laterally compressed. The pollex and dactyl extend beyond the distal margin of the manus; dactyl curving (dorsally) slightly inward (Fig. 8C. d). The manus ventral margin with a beaded ridge is clearest proximally and ends before the pollex base (Fig 8A. e). The outer side of this ridge has a shallow groove on its outer side extending from mid manus to pollex base. Tubercles near the manus dorsal margin become scarce, leaving smooth spaces (Fig. 8D. f); the margin is irregular with a beaded ridge. There is a shallow groove along the outer side of the ridge most evident near the dactyl base (Fig. 8D. g). Manus carpal cavity is squared (Fig. 8B, E. h) and sharply demarcated by a distally continuous, tuberculated edge (Fig. 8B, E. i) that is uniformly rounded and clearly distinct from the flatter manus surface. The upper side of the cavity does not reach the dactylus base (Fig. 8B, E. j). The palm oblique ridge apex starts at the juncture of the distal and lower edges of the carpal cavity (Fig. 8E. k), which is the highest point of the palm; running down to the pollex base with distinct large rounded tubercles (Fig. 8B, E. l). The lower side of the carpal cavity edge is elevated but has no tubercles (Fig. 8E. m). At the pollex base there is a shallow triangular depression that fades distally (Fig 8E. n). The oblique ridge is elevated and forms a triangular projection, located at the center of the palm. The manus inner surface itself is slightly projected upward along the ridge, enhancing the triangular profile (Fig. 8B-C. o). Adjacent to the ridge, the surface is smooth, with only a few small, low tubercles that do not interrupt the overall flatness of the surrounding areas.

Fig. 8. Major cheliped morphological characters of Leptuca bacillopilosa sp. nov. from the Panamanian Pacific. Outer (A), inner (B), and upper view (C) of the major chela surfaces. Structures: a, outer manus tuberculation; b, lower manus surface; c, manus dorsal margin; d, dactylus; e, manus ventral beaded margin; f, sparse tubercles; g, dorsal margin groove; h, carpal cavity; i, distal carpal cavity ridge; j, predactilar upper palm; k, palm apex; l, oblique ridge; m, lower carpal cavity edge; n, pollex base depression; o, triangular palm projection; p, enlarged proximal tubercles; q, dactylar large tooth; r, pollex tricuspid tooth; s, pollex angled tip. Scale = A-C, 1.5 mm; D-E, 2 mm
Fig. 8. Caracteres morfológicos del quelípedo mayor de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Vista externa (A), interna (B) y superior (C) de las superficies de la quela mayor. Estructuras: a, tubérculos externos del manus; b, superficie inferior del manus; c, margen dorsal del manus; d, dáctilo; e, margen ventral del manus con granulación; f, tubérculos dispersos; g, surco del margen dorsal; h, cavidad carpal; i, cresta distal de la cavidad carpiana; j, palma superior predactilar; k, ápice de la palma; l, cresta oblicua; m, borde inferior de la cavidad carpal; n, depresión en la base del pollex; o, proyección triangular de la palma; p, tubérculos proximales agrandados; q, diente grande del dáctilo; r, diente tricúspido del pollex; s, punta angulada del pollex. Escala = A-C, 1.5 mm; D-E, 2 mm
Major dactylus and pollex. The proximal gape is wider than the dactylus, which tapers and curves slightly downward, extending beyond the pollex (Fig. 8A. d). The manus-dactyl articulation cuff is smooth, externally. In large males, the dactyl downward curvature is more pronounced than in smaller males. The dactyl ventral margin starts with three tubercle rows; the outer margin displays small tubercles diminishing in size distally. The mid row contains the largest tubercles; in large individuals, the first to third tubercles may be enlarged (Fig. 8B. p). Approximately halfway along the dactyl, three tubercles fuse, forming a large, round tooth (Fig. 8A. q). The inner row contains three to five small tubercles and fades completely by the first third of the dactyl length. From the cheliped internal view, the pollex dorsal surface bears a short basal row of low, rounded tubercles near the articulation; a second dorsal tubercle row distinct proximally, fading before mid-pollex. There may be pile on either side of the mid tubercle row at the pollex base. In large individuals, the pollex exhibits an enlarged tricuspid tooth arising from the dorsal midline tubercle row (Fig. 8A. r) while the corresponding dactylar tooth is distally offset. In smaller individuals, the dorsal tubercle row persists but remains unfused, with no distinct tooth formation. External pollex surface with a distinct tuberculate ridge extends most of its length, narrowing and fading distally; the tip ends at an angle, with close-set, pointed tubercles forming a rough apex (Fig. 8A. s).
Minor cheliped. The merus is triangular in shape, with weak tubercles along the dorsal margin. The lower inner margin is weakly defined, contrasting with the ventral outer margin, which bears a row of straight, well-defined tubercles. The carpus shows a beaded margin curving subdorsally (Fig. 9A. a). The minor claw is slender, gape narrower than the dactyl and pollex, with the basal third smooth; remainder of the dactyl and dorsal edge of the pollex are moderately serrated (Fig. 9A. b). The outer surface of the pollex has fused tubercles that form a straight margin above the ventral edge, diminishing distally (Fig. 9A. c). Finger tips are spoon-like, bearing long setae distally (Fig. 9B. d).

Fig. 9. Small cheliped morphology of Leptuca bacillopilosa sp. nov. from Panama Pacific. Outer carpus and chela (A), pollex and dactylus tips (B). Structures: a, carpus beaded margin; b, pollex and dactylus serration; c, lower pollex margin; d, distal setae. Scales: A = 1mm; B = 0.5 mm
Fig. 9. Morfología del quelípedo menor de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Cara externa del carpo y quela (A), extremos del pollex y dáctilo (B). Estructuras: a, margen granulado del carpo; b, denticulación del pollex y dáctilo; c, margen inferior del pollex; d, setas distales. Escalas: A = 1 mm; B = 0.5 mm
Ambulatory legs. All meri are broad, with convex dorsal and ventral margins; this is especially pronounced on the third leg in females (Fig. 10A. a). In both sexes, meri edges can be blunt and, in some specimens, they show faint serrations (Fig. 10B. b). On the posterior surface, meri have small tubercles arranged in parallel rows running from the dorsal edge, scattering at the merus midline (Fig. 10B. c). The second and third carpus posterior surfaces, especially in females, and the third in males, bear two anteriorly diverging rows of minute tubercles that run down to the carpus-propodus joint, forming a triangular flat surface (Fig. 10B. d). In males, the anterior surface of the first carpus, on the major side, bears an irregular row of minute tubercles (Fig. 10C. e). Setation is generally absent; pile is sparse, unpatterned, and highly variable, restricted mainly to the posterior surfaces of the meri.

Fig. 10. Ambulatory segment morphology of Leptuca bacillopilosa sp. nov. from the Panamanian Pacific. Posterior meri (A), posterior meri and carpus detail (B), first major side ambulatory surface (C). Structures: a, third posterior meri surface; b, convex upper meri ridge; c, parallel tubercle rows; d, minute tubercle diverging rows; e, minute tubercle irregular row. Scale: A = 2 mm; B = 0.6 mm; C = 1 mm
Fig. 10. Morfología de los pereiópodos ambulacrales de Leptuca bacillopilosa sp. nov. del Pacífico panameño. Superficie posterior de los meros (A), detalle de la superficie posterior de meros y carpo (B), superficie anterior del primer segmento ambulacral (C). Estructuras: a, superficie posterior del tercer mero; b, cresta dorsal convexa del mero; c, hileras paralelas de tubérculos; d, hileras divergentes de tubérculos diminutos; e, hilera irregular de tubérculos diminutos. Escala: A = 2 mm; B = 0.6 mm; C = 1 mm
Maxillipeds. The third maxilliped are robust, with a broad ischium and merus forming a wide plate that covers the buccal region (Fig. 6A, C. d). Dense setation covers the inner margins of both the merus and ischium. The carpus is triangular, short and moderately setose, while the propodus and dactylus are slender and taper distally, bearing scattered simple setae. The second maxillipeds display dense setation along the distal margins of the ischium (Fig. 11A. a), propodus, and dactylus (Fig. 11A. b). Most of the setae are plumose, terminating in flat, pointed tips (Fig. 11A. c), though a few spoon-tipped setae are also present (Fig. 11A. d). These spoon-tipped setae (12-20) are primarily located along the upper mesial margin of the ischium, distinguishable by their broadened, concave distal ends and comb-like margin (Fig. 11B. d).

Fig. 11. Buccal appendage morphology of Leptuca bacillopilosa sp. nov. from Panama Pacific. Second maxilliped setation (A), Setation detail (B). Structures: a, right second maxilliped ischium; b, palpus dactylus; c, plumose flat setae; d, spoon tipped setae. Scale: A = 0.5 mm; B = 0.2 mm
Fig. 11. Morfología de los apéndices bucales de Leptuca bacillopilosa sp. nov. del Pacifico panameño. Setas del segundo maxilípedo (A), detalle de las setas (B). Estructuras: a, isquio del segundo maxilípedo derecho; b, palpo dactilar; c, setas plumosas planas; d, setas punta de cuchara. Escala: A = 0.5 mm; B = 0.2 mm
Reproductive morphology. In males, the abdomen is narrow while in females is wide; in both sexes, the pleonites are asymmetrical, not fused (Fig. 12A-B). The male second pleonite is notably smaller than the other segments (Fig. 12A). The gonopod is curved, without much torsion nor tubercles on the base or shaft. Setae are present at the thumb (Fig. 12C. a) and the proximal surface of the inner process (Fig. 12C. b). The flanges are distinct but underdeveloped (Fig. 12C. c); pore is small, and its internal (anterior) lip is slightly lower than the external, without a projection (Fig. 12C. d). The inner process is moderately broad, flat, nearly reaching the pore (Fig. 12C. e). The thumb is small, derives from a slight basal indentation (Fig. 12C. f), and ends proximal to the corneous base of the pore. Females feature a pair of gonopores, symmetrically positioned at the same level, on either side of the ventral carapace wall (Fig. 12D. g). Each gonopore appears as a depressed, oval cavity (Fig. 12D. h), and the orifice is entirely covered by an operculum (Fig. 12D. i).
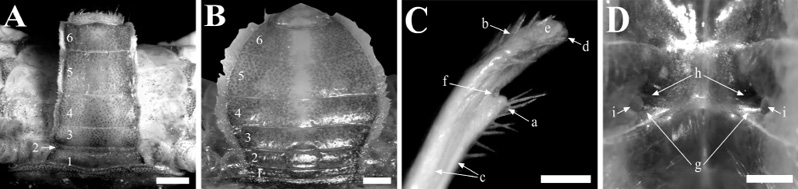
Fig. 12. Leptuca bacillopilosa sp. nov. reproductive morphology from the Panamanian Pacific. Abdomen (A♂-B♀), left gonopod (C♂), gonopores (D♀). Structures: pleonites (1-6); a, thumb setation; b, inner process setation; c, flanges; d, gonopod pore; e, distal inner process; f, thumb basal indentation; g, gonopores; h, ventral carapace wall depression; i, opercula. Scale: A-B = 2 mm; C = 0.1 mm; D = 0.4 mm
Fig. 12. Morfología reproductiva de Leptuca bacillopilosa sp. nov. del Pacifico panameño. Abdomen (A♂-B♀), gonopodio izquierdo (C♂), gonoporos (D♀). Estructuras: pleonitos (1-6); a, setas del pulgar; b, setas del proceso interior; c, juntas; d, poro del gonopodio; e, área distal del proceso interior; f, muesca basal del pulgar; g, gonoporos; h, depresión de pared ventral del caparazón; i, opérculos. Escala: A-B = 2 mm; C = 0.1 mm; D = 0.4 mm
The average CW of L. bacillopilosa in this study (12.53 ± 1.81 mm) places it in the small–medium size range for fiddler crabs, comparable to sympatric species such as L. latimanus (10 - 15.3 mm) but smaller than medium-sized species like L. festae (11 - 21.5 mm; Crane, 1975). Although size is generally a weak criterion for species identification (Crane, 1975; Rosenberg, 2002), the results reveal consistent allometric patterns. This is the case, as front width increases with CW in absolute terms, but its growth is slower, producing a decreasing FW-CW ratio as crabs grow. Furthermore, expressing chela dimensions as ratios yields stronger linear relationships with CW, reflecting predictable, commonplace allometric scaling in fiddler crabs (Rosenberg, 2002).
The recognition of Leptuca bacillopilosa as a distinct species is supported by a suite of diagnostic characters that, in combination, clearly differentiate it from all 18 sympatric congeners (Table 3). A key autapomorphy of L. bacillopilosa is the patterned V-shaped pubescence on the carapace, observed as bacilliform patches in males. Females feature similar pubescence in addition to circular basal patches. This sexually dimorphic arrangement is unique among the sympatric Leptuca, contrasting with either the absence of pile or profuse unpatterned setation in other species (Crane, 1975; Thurman et al. 2025). Additional unique traits such as the strong sigmoidal vertical-lateral margin, and the form of the male gonopod, are not shared with any other of the sympatric species (Crane, 1975; Shih et al. 2016; Rosenberg, 2001; Shih & Chan, 2022).
Beyond singular traits, L. bacillopilosa is distinguished by character combinations. The tapering, downward-curving and inward bending dactylus that extends beyond the pollex, combined with a pollex tricuspid tooth, as well as a large rounded dactylar tooth, yields a configuration unmatched among sympatric taxa. Similarly, the manus displays a unique integration of a keeled ventral margin, a gradient of external manus granulation, and a beaded dorsal ridge. The major cheliped of L. bacillopilosa also bears a steeply sloping oblique ridge with large tubercles that form a triangular projection. This contrasts with the non-projecting ridge of L. terpsichores, the low ridge of L. saltitanta, and the faintly tuberculate ridges of L. oerstedi and L. batuenta, none of which extend to the ventral margin. While L. umbratila shows a ridge that reaches the ventral edge, it is less steep, and in other sympatric congeners the ridge is weak or barely tuberculate. The Caribbean species L. thayeri likewise exhibits only a weak granular ridge (Crane, 1975; Rosenberg, 2019). This distinct ridge profile is therefore of taxonomic value, particularly when considered in concert with the manus traits described above. In the ambulatory legs, broad convex meri and posterior carpal ridges occur in a number of species (Crane, 1975), but only L. bacillopilosa combines these with irregular carpal tuberculation in the first ambulatory, contrasting with its congeners. While many traits are shared within species subsets, such as triangular pollex depressions or triangular minor meri with tubercles, the full array of carapace, cheliped, ambulatory, and gonopod features is distinctive to L bacillopilosa. This integrated diagnostic profile may not be attributed to intraspecific variation of known species. This is the case because intraspecific variation in fiddler crabs, though present in structures such as cheliped size or carapace proportions, is typically limited and does not obscure stable diagnostic features (Hampton et al. 2014; Rosenberg, 2001).
As emphasized in systematic treatments of fiddler crabs (Crane, 1975; Rosenberg, 2001; Shih et al. 2016; Rosenberg, 2019; Beinlich & von Hagen, 2006), reliable species recognition depends not on isolated characters but on stable combinations of morphological traits that persist across populations and geographic ranges (Hampton et al. 2014; Rosenberg, 2002). Accordingly, the integrated set of features in L. bacillopilosa, spanning the dorsal carapace pubescence with bacilliform V-shaped patches in males and sexually dimorphic circular basal pubescence in females, is unique within the genus. In combination with the projecting cheliped triangular tuberculate ridge and distinctive carpal morphology, these traits allow clear differentiation from congeners. The same applies to the ambulatory second and third carpi, where dual diverging beaded ridges are shared with only one congener, and to the distinctive large rounded dactylar tooth and angled, tuberculate pollex tip. These features contrast with the simpler armature of related taxa (Crane, 1975), and together provide a morphologically robust basis for recognizing it as a distinct species.
We thank the anonymous reviewers for their valuable input, which greatly improved the quality of this manuscript. We are also grateful to Michael Rosenberg and Carl Thurman for preliminary discussions, and to Emily Marple and Lara Dominguez for their assistance with English revisions.
Beinlich, B. & von Hagen, H. O. (2006). Materials for a more stable subdivision of the genus Uca Leach. Zool. Meded., 80(4), 9-32.
Booth, J. M., Fusi, M., Marasco, R., Mbobo, T. & Daffonchio, D. (2019). Fiddler crab bioturbation determines consistent changes in bacterial communities across contrasting environmental conditions. Sci. Rep., 9, 3749. https://doi.org/10.1038/s41598-019-40315-0
Bott, R. (1973). Die verwandtschaftlichen Beziehungen der Uca-Arten (Decapoda: Ocypodidae). Senckenb. Biol., 54(4/6), 315-325.
Crane, J. (1941). Eastern Pacific expeditions of the New York Zoological Society. XXVI. Crabs of the genus Uca from the west coast of Central America. Zool. Sci. Contrib. N. Y. Zool. Soc., 26, 145-208.
Crane, J. (1975). Fiddler crabs of the world: Ocypodidae: Genus Uca (1st ed.). EE. UU.: Princeton University Press.
https://doi.org/10.1515/9781400867936
Hampton, K. R., Hopkins, M. J., McNamara, J. C. & Thurman, C. L. (2014). Intraspecific variation in carapace morphology among fiddler crabs (Genus Uca) from the Atlantic coast of Brazil. Aquat. Biol., 20(1), 53-67. https://doi.org/10.3354/ab00545
Lombardo, R. C. (2022). First record of the Fiddler Crab, Minuca osa from the Eastern Montijo Gulf, Panama. Rev. Cienc. Mar. Cost., 14(2), 27-35.
https://doi.org/10.15359/revmar.14-2.2
Lombardo, R. C. (2023). Predation of the fiddler crab, Minuca osa (Brachyura: Ocypodidae), by Eudocimus albus (Pelecaniformes: Threskiornithidae) from Ponuga, Veraguas, Panama. Rev. Cienc. Mar. Cost., 15(2), 2-17.
https://doi.org/10.15359/revmar.15-2.1
Lombardo, R. C. (2024a). New records of Minuca zacae (Brachyura: Ocypodidae) in the Gulfs of Montijo and Parita, Panama. Rev. Cienc. Mar. Cost., 16(1), 33-42. https://doi.org/10.15359/revmar.16-1.2
Lombardo, R. C. (2024b). New records of Leptuca tallanica (Brachyura, Ocypodidae) in the Parita Gulf, Panama, Pacific. Rev. Peru. Biol., 31(3), e28333. https://doi.org/10.15381/rpb.v31i3.28333
Lombardo, R. C. (2025). Inventario de los cangrejos violinista (Brachyura: Ocypodidae) en el Golfo de Parita, Panamá. Scientia, 35(1), 9-54. https://doi.org/10.48204/j.scientia.v35n1.a6621
Mokhtari, M., Ghaffar, M. A., Usup, G. & Cob, Z. C. (2015). Determination of key environmental factors responsible for distribution patterns of fiddler crabs in a tropical mangrove ecosystem. PLoS ONE, 10(1), 17. https://doi.org/10.1371/journal.pone.0117467
Rosenberg, M. S. (2001). The systematics and taxonomy of fiddler crabs: A phylogeny of the Genus Uca. J. Crustac. Biol., 21(3), 839-869. https://doi.org/10.1163/20021975-99990176
Rosenberg, M. S. (2002). Fiddler crab claw shape variation: a geometric morphometric analysis across the genus Uca (Crustacea: Brachyura: Ocypodidae). Biol. J. Linn. Soc., 75(2), 147-162. https://doi.org/10.1046/j.1095-8312.2002.00012.x
Rosenberg, M. S. (2014a). Fiddler crabs. https://www.fiddlercrab.info/index.html
Rosenberg, M. S. (2014b). Contextual cross-referencing of species names for fiddler crabs (Genus Uca): an experiment in cyber-taxonomy. PLoS ONE, 9(7), e101704. https://doi.org/10.1371/journal.pone.0101704
Rosenberg, M. S. (2019). A fresh look at the biodiversity lexicon for fiddler crabs (Decapoda: Brachyura: Ocypodidae). Part 1: Taxonomy. J. Crustac. Biol., 39(6), 729-738. https://doi.org/10.1093/jcbiol/ruz057
Rosenberg, M. S. (2020). A fresh look at the biodiversity lexicon for fiddler crabs (Decapoda: Brachyura: Ocypodidae). Part 2: Biogeography. J. Crustac. Biol., 40(4), 365-383. https://doi.org/10.1093/jcbiol/ruaa029
Shih, H. Te, Ng, P. K. & Christy, J. H. (2015). Uca (Petruca), a new subgenus for the rock fiddler crab Uca panamensis (Stimpson, 1859) from Central America, with comments on some species of the american broad-fronted subgenera. Zootaxa, 4034(3), 471-494. https://doi.org/10.11646/zootaxa.4034.3.3
Shih, H. Te., Ng, P. K., Davie, P. J., Schubart, C. D., Türkay, M., Naderloo, R., ... & Liu, M. Y. (2016). Systematics of the family Ocypodidae Rafinesque, 1815 (Crustacea: Brachyura), based on phylogenetic relationships, with a reorganization of subfamily rankings and a review of the taxonomic status of Uca Leach, 1814, sensu lato and its subgenera. Raffles Bull. Zool., 64, 139-175.
Shih, H. Te. & Chan, B. K. (2022). Systematics and biogeography of fiddler crabs – A special issue in zoological studies. Zool. Stud., 61(64), 1-5. https://doi.org/10.6620/zs.2022.61-64
Thurman, C. L., McNamara, J. C., Shih, H. Te. & Capparelli, M. V. (2025). Fiddler crabs (Crustacea: Decapoda: Ocypodidae) from coastal Ecuador and the Galápagos Islands: Species descriptions and DNA barcodes. Ecol. Evol., 15(1), e70646. https://doi.org/10.1002/ece3.70646
von Hagen, H. O. (1968). Studien an peruanischen winkerkrabben (Uca). Zool. Jahrb. Syst., 95(2), 395-468.
Xu, X., He, L., Huang, F., Jiang, S., Dai, Z., Sun, R. & Li, C. (2024). Fiddler crabs (Tubuca arcuata) as bioindicators of microplastic pollution in mangrove sediments. Chemosphere, 364, 143112. https://doi.org/10.1016/j.chemosphere.2024.143112
1 Universidad de Panamá, Departamento de Biología Marina, Centro Regional Universitario de Veraguas.
2 Centro de Capacitación, Investigación y Monitoreo de la Biodiversidad en el Parque Nacional Coiba.
3 Sistema Nacional de Investigación, Secretaría Nacional de Ciencia, Tecnología e Innovación, Panamá.
roberto.lombardo@up.ac.pa* ORCID: https://orcid.org/0000-0002-0279-8621




