ISSN: 1659-455X • e-ISSN: 1659-407X
Vol. 17 (2), julio-diciembre 2025
Recepción 30 septiembre 2024 • Corregido 16 enero 2025 • Aceptado 16 junio 2025
DOI: https://dx.doi.org/10.15359/revmar.17-2.1
 |
Antioxidant capacity of hydroethanolic extracts from Thalassia testudinum and Syringodium filiforme to counteract UVB radiation effects on skin Capacidad antioxidante de los extractos hidroetanólicos de Thalassia testudinum y Syringodium filiforme para contrarrestar los efectos de la radiación UVB en la piel Adrián Fagundo-Mollineda1*, Ruth Ana Morales-Aguilera2, Roberto Menéndez-Soto del Valle3, Kethia González-García2, Teidy García-Jiménez2, Olga Valdés-Iglesia2, |
ABSTRACT
The prevalence of skin diseases related to ultraviolet radiation (UV) from the sun has increased alarmingly. Direct exposure to UV rays significantly reduces the levels of endogenous antioxidants in the skin. The accelerated increase of free radicals triggers the activation of inflammatory processes that damage collagen and elastin fibers, leading to faster photoaging and the long-term development of melanoma-like lesions. However, searching for new natural components and extracts that can restore endogenous antioxidant levels through topical applications offers a promising avenue for effective treatments. This study aims to evaluate the ability of extracts obtained from the marine angiosperm Thalassia testudinum Banks & Sol. ex Koenig and Syringodium filiforme Kützing in Hohenacker to modify oxidative stress markers (MDA, GSH, SOD) in an in vivo model of UVB light-induced skin photodamage. The leaves of both plants were collected in Rincón de Guanabo, Cuba, in November 2022. Both hydroethanolic extracts have a high content of phenolic compounds (18 ± 1.5 and 25.78 ± 0.07 PyE%) and other natural antioxidants that inhibit lipid peroxidation with IC-50 below 0.008 mg/mL. Results also demonstrate the antioxidant capacity of both extracts in restoring oxidative stress indicators, including MDA, GSH, and SOD values, closer to those found in healthy, non-irradiated skin. The extract of T. testudinum has a greater capacity to increase intracellular SOD enzyme levels. The present research suggests that topical treatment may contribute to maintaining antioxidant levels in the skin, thereby reducing the harmful effects of oxidative imbalance and offering a promising prospect for effective therapies.
Keywords: Thalassia, Syringodium, UVB radiation, antioxidant activity, SOD
RESUMEN
La prevalencia de enfermedades de la piel relacionadas con la radiación solar ultravioleta (UV) ha aumentado de forma alarmante. La exposición directa a los rayos UV reduce los niveles de antioxidantes endógenos en la piel. El aumento acelerado de radicales libres puede activar procesos inflamatorios que dañan las fibras de colágeno y elastina, provocando un fotoenvejecimiento acelerado y la aparición de lesiones tipo melanoma, a largo plazo. Sin embargo, la búsqueda de nuevos componentes y extractos naturales que puedan restaurar los niveles de antioxidantes endógenos, mediante aplicaciones tópicas, ofrece esperanzas de tratamientos efectivos. El trabajo tiene como objetivo evaluar la capacidad de los extractos obtenidos de las angiospermas marinas Thalassia testudinum y Syringodium filiforme de modificar los indicadores de estrés oxidativo en un modelo de fotodaño cutáneo inducido con luz UVB. Las hojas de ambas plantas se recolectaron en Rincón de Guanabo, Cuba, en noviembre de 2022. Ambos extractos presentan alto contenido de polifenoles (18 ± 1.5 y 25.78 ± 0.07 PyE%) y otros antioxidantes que inhiben la peroxidación lipídica con IC-50 menores 0.008 mg/mL. Los resultados demuestran, además, la capacidad antioxidante de ambos extractos para reestablecer los indicadores de estrés oxidativo: MDA, GSH y SOD hacia valores cercanos a los encontrados en pieles sanas no irradiadas. El extracto de T. testudinum presenta una mayor capacidad para aumentar los niveles de enzima SOD intracelular. La presente investigación sugiere que el tratamiento tópico podría contribuir a mantener los niveles de antioxidantes en la piel, reduciendo los efectos nocivos del desequilibrio oxidativo.
Palabras clave: Thalassia, Syringodium, Radiación UVB, Actividad antioxidante, SOD
Four decades ago, researchers in the UK uncovered the damage to the ozone layer. Gas concentration above the Antarctic sky plummeted below the global historical record of 220 Dobson units (Newman et al. 2006). UV radiation is classified into three categories according to wavelength: UVA (320-400 nm), UVB (280-320 nm), and UVC (200-280 nm). UV-B radiation is highly damaging and is inversely related to total ozone: the lower the amount of gas, the greater the penetration of UV-B radiation into the Earth’s surface (McKenzie et al. 2003). Recently, skin diseases related to ultraviolet solar radiation have increased dramatically (Lucas et al. 2019; Autier & Doré, 2020; Parker, 2021), underscoring the urgent need to search for new compounds to mitigate their harmful effects.
The skin is the most exposed organ to the harmful effects of UV-B radiation; the stratum corneum, the outermost layer of the epidermis, absorbs approximately 70% of the UV-B radiation. The human body has developed various actinic self-protection mechanisms, such as the formation of melanin, increased stratum corneum thickness, and the secretion of sweat containing urocanic acid. However, the damage could be irreversible if exposure duration, frequency, and intensity exceed the tissue’s defensive capacity. Actinic erythema, photoaging, and skin cancer are among the most common photobiological consequences (Lucas et al. 2019).
Topical application of antioxidants to the skin, as part of commercial cosmetic products, may be an effective photoprotection strategy (Hoang et al. 2021). In this way, conventional passive sunscreens that absorb or reflect UV-B wavelengths can be combined with biologically active components to mitigate the significant decrease in endogenous antioxidant levels that accompanies prolonged exposure to this radiation (Jesus et al. 2023). Using these cosmeceuticals, which possess medicinal properties, has beneficial local effects and helps prevent degenerative skin diseases (Husein & Castillo, 2016).
The Cuban insular shelf is covered by many marine seagrass beds, among which the marine angiosperm species Thalassia testudinum and Syringodium filiforme are abundant (Martínez-Daranas & Suárez, 2018). Their high density in the region, combined with an efficient collection method, allows for their sustainable exploitation and offers ample opportunities to select plants for searching and exploiting bioactive products and molecules without causing damage to the ecosystem or disrupting their functions in the environment where they grow.
T. testudinum and S. filiforme are organisms that develop in complex ecological conditions and are sensitive to variations in the marine environment (Wuthirak et al. 2016). Tidal changes expose these organisms to dehydration and high temperatures during low tide due to direct sunlight (Phandee & Buapet, 2018). These factors, combined with marine and anthropogenic pollution in the area, contribute to oxidative stress in the tissues of these plants, thereby increasing the levels of reactive oxygen species (ROS) within the cells (Fagundo-Mollineda et al. 2023). This large amount of ROS can cause damage to significant macromolecules, including lipids, proteins, and nucleic acids. However, to counteract extreme oxidation, marine angiosperms produce a series of enzymatic and non-enzymatic components that can be used to maintain oxidative balance within the cell. Most of these metabolites benefit humans and can be isolated and used as excellent therapeutic products (Fagundo-Mollineda et al. 2023). Recent studies have demonstrated that the extracts of these marine macrophytes exhibit significant antioxidant activity, primarily due to their polyphenolic content (Halliwell, 2008; Jafriati et al. 2019).
Although not extensively studied as a source of bioproducts, seagrasses have shown potential applications in treating various disorders due to their antioxidant properties (Perry et al. 2024). In this sense, Regalado et al. (2012) and González et al. (2011; 2017) demonstrated that hydroethanolic extracts obtained from T. testudinum and S. filiforme possess, in addition to a high content of polyphenols and other natural antioxidants, an in vitro capacity for the elimination of free radicals. Hernández-Balmaseda et al. (2021) found that the extract of T. testudinum can suppress the growth, motility, and angiogenesis of colorectal tumors. Kevrekidou et al. (2024) propose that extracts of Posidonia oceanica exhibit antioxidant and antitumor activity due to their high phenolic content in both live plants and beach debris. Among the main compounds isolated from seagrasses with bioactive potential, we can mention thalassiolins B and D, which are present in T. testudinum and Thalassia hemprichii (Regalado et al. 2009; Hawas & Abou El-Kassem, 2017). The presence of polyphenols, terpenes, and phenylheptanoids, as well as p-hydroxybenzoic, coumaric, and caffeic acids, is suggested as the most common secondary metabolites (Zidorn, 2016).
When analyzing antioxidant activity, authors frequently employ combinations of in vitro analytical methods, such as DPPH, FRAP, ABTS, and CUPRAC, which can accurately, quickly, and inexpensively quantify this activity (Munteanu & Apetrei, 2021). However, few studies using in vivo experimental models allow us to understand how antioxidant molecules and enzymes function in target organs, such as the skin. Based on this background, the objective of this study was to evaluate the antioxidant capacity of extracts obtained from the marine angiosperms Thalassia testudinum and Syringodium filiforme to modify oxidative stress markers (malondialdehyde (MDA), glutathione (GSH), and superoxide dismutase (SOD)) in an in vivo model of UVB-induced skin photodamage.
Specimens of the species T. testudinum and S. filiforme were collected by apnea at a depth between 1 and 3 meters in Rincón de Guanabo, Havana, Cuba (23° 11ʼ 12ʼʼ N and 82° 06ʼ 28ʼʼ W) in November 2022. Only the leaves of both species were harvested to minimize the impact on their ecosystem functions and clonal reproductive strategies. The samples were washed, drained, and dried at 60 °C in a forced-air oven. One specimen of each species was placed in the collection of specimens at the Herbarium of the National Aquarium of Cuba (IDO 169 and IDO 177).
Extraction: Two hundred grams of dry, powdered plant material (0.12 mm) were macerated in a 50% (vol/vol) ethanol-water solution from each species using mechanical stirring for seven days at room temperature. After filtration and vacuum concentration, the samples were prepared for the treatments.
Antioxidant characterization of the extracts by in vitro methods
Preparation of brain tissue homogenates
A brain tissue homogenate served as an in vitro experimental model for membranes in animal tissue, acting as a source of phospholipids to induce spontaneous lipid peroxidation. To obtain the brain homogenates, albino mice (males weighing between 20 and 25 g) were used from the OF-1 line. They were obtained from the National Center for the Production of Laboratory Animals (CENPALAB), Havana. Animals were previously anesthetized in an ether atmosphere and euthanized by cervical traction. Homogenates were prepared in a 10 mM phosphate-buffered saline solution (PBS) at pH 7.4, using a w/v ratio of 1:9, with a Potter–Elvehjem type manual homogenizer immersed in an ice bath. Subsequently, the samples were centrifuged at 4000 g for 20 min at 0-4°C and stored frozen until the lipid peroxidation assay was performed.
Assay for determining lipid peroxidation (LPO) in brain homogenate
The inhibition of spontaneous lipid peroxidation in the homogenates was measured using the method described by Cini et al. (1994). To achieve this, samples were incubated at a concentration of 500 µg/mL for 30 minutes at 37°C in the presence of extracts at 0.5 mg/mL. In parallel, control tubes received an equivalent volume of the buffer used in the procedure, while positive control tubes received the same volume of butylated hydroxytoluene (BHT) at the test concentrations. The degree of peroxidation in the different samples was determined spectrophotometrically by forming reactive thiobarbituric acid substances (TBARs) (Ohkawa et al. 1979) at a wavelength of 532 nm. The extract concentration required to inhibit 50% of the maximum formation of TBA-reactive substances (IC-50) was determined.
Quantification of total polyphenol content
Total phenolic compounds were quantified following the Folin & Ciocalteu (1927) methodology modified by Lim et al. (2002). A total of 200 μL of extract was combined with 800 μL of Folin-Ciocalteu reagent and 200 μL of 29% sodium carbonate (Na2CO3). The mixture was incubated at room temperature for 30 minutes, and the absorbance was measured at 760 nm. The total phenol content was calculated using a Pyrogallol standard curve and expressed as a percentage of Pyrogallol equivalents (PyE%).
Determination of antioxidant effects in the in vivo model of photodamage induced by UVB light in murine skin
Animals and treatments: The experiments were conducted using male albino mice of the Balb/c line (22-24 g body weight) from the Center for the Production of Laboratory Animals, Havana. Animals were provided with pelleted feed (CENPALAB), water, and food ad libitum, ensuring their well-being. They were kept in quarantine for seven days before the start of the experiments, a standard practice to prevent any potential health issues. Animals were treated with the utmost respect and care, in accordance with institutional guidelines for working with laboratory animals, as outlined in the Declaration of Helsinki (2020).
Forty-eight hours before the test, the dorsal area (2 x 2 cm) of the mice was depilated using Veet cream (France). Mice were then divided into five experimental groups, each consisting of ten animals. (Experimental groups: Control: Animals with healthy, unirradiated skin; Irradiated: Animals with irradiated skin that received no treatment; Placebo: Animals with irradiated skin plus a vehicle; ThtE: Animals with irradiated skin plus T. testudinum extract; SyfE: Animals with irradiated skin plus S. filiforme extract). Before radiation exposure, they were anesthetized with sodium pentobarbital (83.3 mg/kg body weight). According to the Photodamage Model (Regalado et al. 2009), mice were exposed to ultraviolet B radiation (312 nm) with a Spectroline lamp (Spectronics). Irradiance was focused on the back of the mice (30 cm from the lamp) for 6 minutes at 0.3 mW/cm². Immediately afterward, treatment with the study products commenced. After 72 hours, euthanasia was performed using pentobarbital sodium intraperitoneally (100 mg/kg), and skin samples were collected for tests to determine the antioxidant action of the extracts on the skin (Fig. 1).
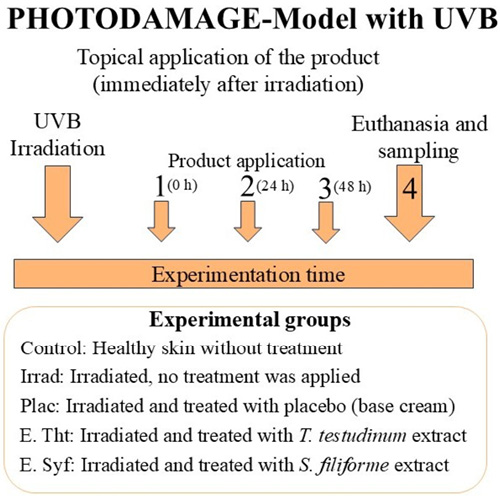
Fig. 1. Experimental diagram and treatment group used in the photodamage before studying the antioxidant activity
Fig. 1. Diagrama experimental y grupos de tratamientos utilizados en el modelo fotodaño antes de estudiar la actividad antioxidante
Crude extracts of T. testudinum and S. filiforme were manually mixed with the vehicle to create a homogeneous cream (0.008 mg of crude extracts per milligram of vehicle (w/w)). The base cream used as a vehicle was a simple oil-in-water cosmetic emulsion, formulated without preservatives and with an equal volume of added distilled water. Aliquots of the cream were applied evenly over the dorsal area using an applicator at a concentration of 500 µg/cm². Mice were kept isolated for approximately 15 minutes to prevent the cream from being contaminated by contact with surrounding surfaces.
Preparation of skin homogenates
Skin samples were taken from all the animals to obtain the homogenates. The pieces were previously washed in physiological saline solution (0.9% NaCl) and carefully cut with scissors. Next, they were homogenized in a solution of phosphate-buffered saline (PBS) at pH 7.4, with a w/v ratio of 1:9, using a manual “potter”-type homogenizer submerged in an ice bath. Subsequently, the samples were centrifuged at 0-4°C and then frozen until the various tests were performed.
Determination of malondialdehyde content
MDA constitutes one of the final products of lipid peroxidation and represents a measure of the extent of damage caused by oxidation of the cell membrane. The metabolite content was determined by modifying the methodology proposed by Ohkawa et al. (1979). The concentrations of reactive thiobarbituric acid substances were determined using the molar extinction coefficient of the MDA-TBA complex (1.56 × 105 M-1 cm-1) and expressed in units of molarity per concentration.
Determination of reduced glutathione level
GSH levels were quantified using Ellman’s reagent (DTNB 5,5’-Dithio-bis-(2-nitrobenzoic acid), which reacts with the sulfhydryl groups (–SH) present in the mixture, producing a yellow solution that absorbs at a wavelength of 412 nm (Owen & Butterfield, 2010). The concentration of thiol groups was determined based on the molar extinction coefficient of DTNB (14.150 M-1 cm-1). Values were referred to the concentration of cellular proteins. The protein concentration was determined using a standard curve prepared with bovine serum albumin (BSA), and data were obtained from this curve.
Determination of cytosolic superoxide dismutase enzyme levels
The determination of the enzymatic activity is based on the methodology described by Marklund & Marklund (1974). Pyrogallol auto-oxidizes in an essential medium, generating superoxide radicals in the reaction medium. In this way, the radical reaction propagates, accelerating the oxidation of Pyrogallol, whose oxidized form absorbs light at 420 nm. The enzyme SOD inhibits auto-oxidation by dismutation of the superoxide radical (O2•-) to hydrogen peroxide (H2O2). 1 U of SOD (amount of enzyme that inhibits the autoxidation of Pyrogallol by 50%, 25°C, pH: 8.20). The activity values are referred to as protein concentrations. The protein concentration was determined using a standard curve prepared with bovine BSA and data obtained from this curve.
Statistical analysis of the results
Statistical analysis was performed using SPSS 27.0 (IBM Corp., 2020). To select the statistical methods, tests of data normality (Kolmogorov-Smirnov test) and homogeneity of variance (Bartlett test) were performed. The means of the data obtained were compared using simple classification analysis of variance (ANOVA), followed by Tukey’s multiple comparison tests. Probabilistic values (P) less than 0.05 were considered statistically significant.
Antioxidant characterization of the extracts by in vitro methods
Hydroethanolic extracts of T. testudinum and S. filiforme contained high levels of phenolic compounds, 18.0 ± 1.5 PyE% and 25.8 ± 0.1 PyE%, respectively. Furthermore, both extracts effectively inhibited lipid peroxidation. T. testudinum extract inhibited spontaneous lipid peroxidation in vitro with an IC-50 of 0.006 mg/mL, whereas the mean inhibitory concentration for S. filiforme extract was 0.008 mg/mL.
Analysis of malondialdehyde (MDA) concentrations in the photodamage model
As shown in Figure 2, both the irradiated and placebo groups showed a significant increase in malondialdehyde (MDA) levels compared to the control group. In animals treated with the extracts, MDA content decreased compared to the other irradiated animals, suggesting that the extracts inhibit the propagation of lipid peroxidation. No significant differences were observed between the animals treated with the extracts.
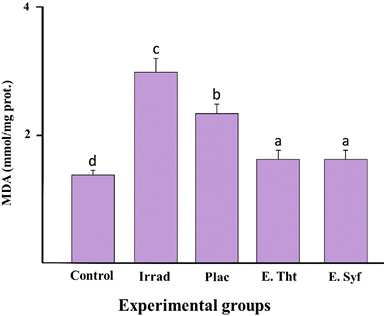
Fig. 2. Comparison of MDA concentrations in the photodamage model. Control: Healthy skin; Irrad: Irradiated without treatment; Placebo: Irrad + Vehicle; E.Tht: Irrad + Extract of T. testudinum; E. Syf: Irrad + Extract of S. filiforme. Dosage: 500 μg.cm-2. Different letters indicate significant differences between groups. Tukey tests, P ≤ 0.05 n=10, lg=4
Fig. 2. Comparación de concentraciones de MDA en el modelo de fotodaño. Control: Piel sana; Irrad: Irradiado sin tratamiento; Placebo: Irrad + Vehículo; E.Tht: Irrad + Extracto de T. testudinum; E.Syf: Irrad + Extracto de S. filiforme. Dosis: 500 μg.cm-2. Letras diferentes indican diferencias significativas entre grupos. Pruebas de Tukey, P ≤ 0.05 n=10, gl=4
Analysis of the concentrations of GSH in the photodamage model
When analyzing GSH concentrations in the various experimental groups, we found that both the irradiated and placebo groups exhibited significantly lower GSH levels compared to the control group. In the groups treated with the extracts, a significant increase in total thiol concentrations was observed, comparable to the levels reached in the control group (Fig. 3). Furthermore, the groups receiving the extracts displayed statistically similar behavior.
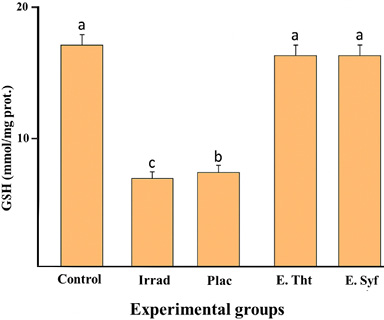
Fig. 3 Comparison of GSH concentrations in the photodamage model: Control: Healthy skin; Irrad: Irradiated without treatment; Placebo: Irrad + Vehicle; E.Tht: Irrad + Extract of T. testudinum; E.Syf: Irrad + Extract of S. filiforme. Dosage: 500 μg.cm-2. Different letters indicate significant differences between groups. Tukey tests, P ≤ 0.05. n = 10, lg=4
Fig. 3 Comparación de concentraciones de GSH en el modelo de fotodaño Control: Piel sana; Irrad: Irradiado sin tratamiento; Placebo: Irrad + Vehículo; E.Tht: Irrad + Extracto de T. testudinum; E.Syf: Irrad + Extracto de S. filiforme. Dosis: 500 μg.cm-2. Letras diferentes indican diferencias significativas entre grupos. Pruebas de Tukey, P ≤ 0.05. n = 10, gl=4
Analysis of the activity of the enzyme superoxide dismutase (SOD) in the photodamage model
Analysis in Figure 4 indicates that both the irradiated and placebo groups showed a significant reduction In SOD enzyme activity compared to the control group. In contrast, the other groups showed results that were opposite to these. The groups treated with the extracts showed more significant SOD enzyme activity, particularly the group treated with the T. testudinum extract, which was significantly different from the other groups.
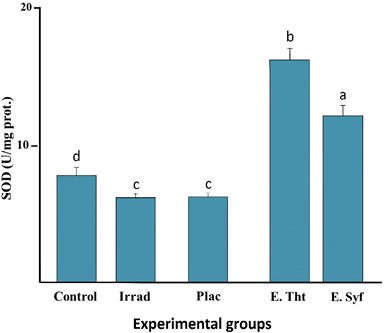
Fig. 4. Comparison of SOD enzyme activity in the photodamage model Control: Healthy skin; Irrad: Irradiated without treatment; Placebo: Irrad + Vehicle; E.Tht: Irrad + Extract of T. testudinum; E.Syf: Irrad + Extract of S. filiforme. Dosage: 500 μg.cm-2. Different letters indicate significant differences between groups. Tukey tests, P ≤ 0.05. n = 10, lg=4
Fig. 4. Comparación de la actividad de la enzima SOD en el modelo de fotodaño Control: Piel sana; Irrad: Irradiado sin tratamiento; Placebo: Irrad + Vehículo; E.Tht: Irrad + Extracto de T. testudinum; E.Syf: Irrad + Extracto de S. filiforme. Dosis: 500 μg. cm². Letras diferentes indican diferencias significativas entre grupos. Pruebas de Tukey, P ≤ 0.05. n = 10, gl=4
In most studies, a high concentration of phenolic compounds is associated with a notable antioxidant activity (Zubia et al. 2007; Fagundo-Mollineda et al. 2023). When working with crude hydroethanolic extracts, many natural molecules may coexist in the mixture, collaborating to produce a synergistic anti-free radical effect. This creates an effective defense system against cellular damage that encompasses all possible routes of formation and propagation (Lim et al. 2022).
As we can see in the results, the T. testudinum extract, despite presenting a lower amount of phenolic compounds, is capable of inhibiting spontaneous lipid peroxidation at a lower extract concentration; this may be due to the reported presence in said mixture of very potent antioxidant molecules such as thalassiolin B (Regalado et al. 2009), a sulfate ester, and ten phenolic derivatives (Regalado et al. 2012). The thalassiolin B molecule was isolated from a T. testudinum extract and tested as a topical antioxidant in a model similar to the one used in this study. The results showed that continuous administration of thalassiolin B to photodamaged skin significantly reduced skin damage caused by UVB radiation exposure (Regalado et al. 2009).
From a physiological point of view, even though both angiosperms share a habitat and are exposed to the same environmental conditions (amount and quality of light, temperature, salinity, pollution), the morphology of T. testudinum means that its leaves are more exposed to UV radiation because they have a larger contact surface area and therefore need to produce a greater variety of antioxidant compounds to reestablish their redox balance and eliminate free radicals (Fig. 5).
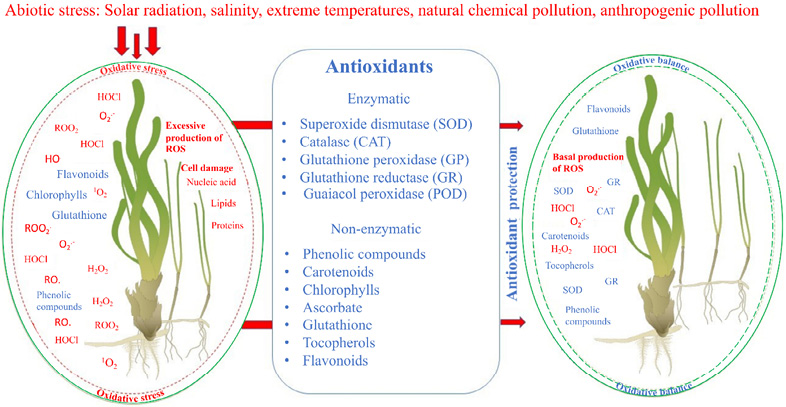
Fig. 5. Main sources of oxidative stress, ROS, and antioxidants in seagrasses. This scheme represents the interior of a plant cell from an oxidative perspective, highlighting the main factors associated with oxidative stress versus oxidative balance. ROS: O2.- superoxide anion, H2O2: Hydrogen peroxide, HO.: Hydroxyl radical, ROO2.: Peroxyl radical, RO.: Alkoxyl radical, ROOH.: Hydroperoxide, 1O2: Oxygen singlet, HClO: Hypochlorous acid
Fig. 5. Principales fuentes de estrés oxidativo, ROS y antioxidantes en praderas marinas. Este esquema representa el interior de la célula vegetal desde el punto de vista oxidativo, representando los principales factores asociados al estrés oxidativo frente al balance oxidativo. ROS: O2.- anión superóxido, H2O2: Peróxido de hidrógeno, HO.: Radical hidroxilo, ROO2.: Radical peroxilo, RO.: Radical alcoxilo, ROOH.: Hidroperóxido, 1O2: Singlete de oxígeno, HClO: Ácido hipocloroso
Previous studies have reported the antioxidant activity present in extracts of T. testudinum and S. filiforme related to their ability to neutralize free radicals associated with lipid peroxidation. Regalado et al. 2012 reported that the extract of T. testudinum can neutralize hydroxyl radicals, superoxide, and peroxyl, which are the main initiators of lipid peroxidation. Although the hydroethanolic fraction of S. filiforme has not been very characterized, González et al. (2011) state that in their analysis, the total extract and the methanolic fraction obtained from S. filiforme showed important antioxidant properties using the DPPH method.
Regarding the ability of our extracts to inhibit spontaneous lipid peroxidation in vitro, other authors have found similar effects using plant extracts containing polyphenols or evaluating pure polyphenolic compounds. Cheung and Cheung (2005) demonstrated that different fractions obtained from methanolic and aqueous extracts of the mushrooms Lentinus edodes (shiitake) and Volvariella volvacea (Straw mushroom) inhibited LPO in the same experimental model used in the present work. Other studies have highlighted the antioxidant properties of the aqueous extract of the red alga Alsidium triquetrum, which effectively inhibits spontaneous LPO in brain homogenates, exhibiting an IC-50 value of 0.800 mg/ mL (Fallarero et al. 2006) and extracts of similar nature from the green algae Halimeda incressata and Halimeda monile, which inhibit spontaneous LPO in brain tissue, exhibiting an IC-50 value of 0.340 mg/ mL (Rivero et al. 2003) and 2.19 mg/ mL (Batista-Gonzalez et al. 2012), respectively. Most of these values are higher than those found in our study, indicating a lower LPO inhibitory power in these extracts, which lends relevance to the antioxidants present in the extracts of T. testudinum and S. filiforme.
MDA is one of the products generated during the lipid oxidation process in biological membranes, capable of causing structural alterations that lead to a loss of fluidity, thereby increasing their rigidity and instability. For this reason, it is widely used to monitor the level of lipoxidation in cellular membranes due to its high reactivity and toxicity (Mas-Bargues et al. 2021). MDA is an end product formed by decomposing arachidonic acid and other polyunsaturated fatty acids (PUFAs). Excessive MDA production has been associated with several pathological states, including Alzheimer’s disease, cancer, cardiovascular disease, diabetes, and liver disease (Ayala et al. 2014).
Our trials showed that MDA levels in the untreated irradiated animal groups increased significantly above those in animals treated with T. testudinum and S. filiforme extracts. This suggests that the extracts have the potential to inhibit the progression of lipid peroxidation in irradiated skin.
Lephart (2016) and Mitchell (2016) suggest that UVB light interacts with the unsaturated phospholipids that comprise the biological cellular membranes within the photodamaged area, resulting in the formation of new free radicals. Through propagation, these free radicals promote lipid peroxidation. The lipoperoxides that are formed, in turn, induce the oxidation of collagen and elastin and can damage various molecules, including deoxyribonucleic acid (DNA). Furthermore, the free radicals that continue to form oxidize the lipids present in the newly formed tissue, contributing to the continued rise in MDA concentrations if an effective treatment is not applied (Ayala et al. 2014).
In a previous study by Fagundo-Mollineda et al. (2016) on these extracts, it was observed that untreated skin showed histopathological alterations such as acanthosis (thickening of the epidermis) due to hyperplasia and hypertrophy of epidermal cells, hyperkeratosis (thickening of the stratum corneum) due to keratin accumulation, infiltration of inflammatory cells and congestion of blood vessels. However, in animals treated with T. testudinum and S. filiforme extracts, these alterations were significantly reduced to a level comparable to that of non-irradiated healthy skin, indicating that the damage does not persist and that the extracts slow the spread of lipid peroxidation.
According to Gupta et al. (2002), 72 hours after irradiation, a decrease in endogenous antioxidant concentrations can be expected in the skin tissue of animals exposed to UVB light, compared to the values of these indicators in intact skin. This moment corresponds to the inflammatory phase of the wound healing process. This phase is characterized by the infiltration of polymorphonuclear leukocytes and macrophages into the injured tissue, which, while phagocytizing cell fragments and debris from damaged tissue, produces a large amount of ROS (Sen, 2003; Sen, 2009). Therefore, endogenous antioxidant sources are utilized to neutralize newly generated ROS, thereby maintaining cellular redox balance.
SH group functions as an electron donor and is responsible for the biological activity of reduced glutathione. The irradiated and placebo groups exhibited significantly lower GSH levels compared to the control group, indicating that the degree of damage persisted for 72 hours after the induced phase (Fig. 4). This may be because endogenous antioxidants are depleted or reach low levels in the skin, and the synthesized glutathione is rapidly oxidized to reduce oxidative damage. It is fascinating to observe how this alteration is not maintained in the remaining groups. In the groups treated with the extracts, a significant increase in total thiol concentrations was observed, comparable to the levels achieved in non-irradiated skin. This analysis demonstrated that both extracts reduce oxidative stress in the skin, enabling GSH levels to return to typical values within 72 hours. This is particularly important when considering the crucial role that reduced glutathione plays as a component of the cellular redox system (Averill-Bates, 2023), which reinforces the significance of our findings.
Cells can regulate their antioxidant system, which comprises both enzymatic and non-enzymatic components (Fig. 5). One of the main enzymes reported to have this property is SOD. In our study, both the irradiated and placebo groups exhibited a significant reduction in activity compared to the control group. This may be due to the damage that persists after 72 hours of exposure to UVB light. However, the other groups showed opposite results.
The groups treated with the extracts exhibited a more significant activity of the SOD enzyme, with the group treated with the T. testudinum extract showing significant differences compared to the other groups. A possible explanation for these findings could be the possible activating effect of the extracts on the enzymes that make up the endogenous antioxidant system of the skin, considering the high content of polyphenols that both possess and the presence of the significant components of the T. testudinum extract, the glycosylated flavone thalassiolin B (Regalado et al. 2009). Flavones behave as a potent inducer of antioxidant enzymes (Regalado et al. 2012), which coincides with the fact that in the group treated with the T. testudinum extract, the SOD activity increases to levels significantly different from the control. However, it is necessary to point out that, despite the numerous experimental evidence suggesting that the activation of the endogenous antioxidant system is incorporated into the vast arsenal of pharmacological activities of polyphenols, the molecular mechanisms through which these increases are induced have not been fully elucidated.
The results obtained in the present investigation demonstrate that both extracts significantly reduce the concentration of MDA, an indirect marker of membrane damage, and increase the concentration of GSH and the activity of the SOD enzyme to a significant level in the selected in vivo model. Therefore, one possible mechanism of protection against damage induced by UVB radiation in murine skin may be closely linked to its antioxidant action (Svobodová et al. 2003). Thus, the extracts of both angiosperms would contribute to limiting the oxidative imbalance initiated by the decrease in GSH concentrations, avoiding, on the one hand, the increase of ROS in skin tissue and, on the other hand, the maintenance of endogenous antioxidant defenses, which together would slow the increase in POL and its deleterious effects on the skin.
The extracts of Thalassia testudinum and Siringodium filiforme inhibit spontaneous lipid peroxidation in vitro in brain homogenate and have a high percentage of total polyphenols. Additionally, our extracts reduce MDA concentrations and increase the activity of the antioxidant enzyme SOD, as well as the content of total thiol groups (GSH), in skin tissue. Both extracts, especially those of T. testudinum, constitute an essential source of marine bioproducts of pharmacological interest for skincare products. Therefore, we recommend continuing to work on their characterization, analysis, and purification.
We sincerely thank María Iglesias-Rodríguez for her meticulous support in editing the final document. We want to express our gratitude to the anonymous reviewers for their valuable comments, which significantly improved the quality of the manuscript.
Autier, P. & Doré, J. F. (2020). Ultraviolet radiation and cutaneous melanoma: a historical perspective. Melanoma Res., 30(2), 113-125. https://doi.org/10.1097/CMR.0000000000000609
Averill-Bates, D. A. (2023). The antioxidant glutathione. In G. Litwack (Ed.), Vitamins and hormones (pp. 109-141). Academic Press. https://doi.org/10.1016/bs.vh.2022.09.002
Ayala, A., Muñoz, M. F. & Argüelles, S. (2014). Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev., 2014(1), 360438. https://doi.org/10.1155/2014/360438
Batista-Gonzalez, A. E., Silva, A., Vidal-Novoa, A., Pinto, J. R., Mancini, D. A. P. & Mancini-Filho, J. (2012). Analysis of in vitro and in vivo antioxidant properties of hydrophilic fractions from the seaweed Halimeda monile L. J. Food Biochem., 36(2), 189-197. https://doi.org/10.1111/j.1745-4514.2010.00525.x
Cheung, L. M. & Cheung, P. C. (2005). Mushroom extracts have antioxidant activity against lipid peroxidation. Food Chem., 89(3), 403-409. https://doi.org/10.1016/j.foodchem.2004.02.049
Cini, M., Fariello, R. G., Bianchetti, A. & Moretti, A. (1994). Studies on lipid peroxidation in the rat brain. Neurochem. Res., 19, 283-288. https://doi.org/10.1007/BF00971576
Fagundo-Mollineda, A., García, M. R., García, K. L. G., Iglesias, O. V., Rivera, Y. H., Alfonso, Á. R. C. & Martínez, O. V. (2016). Actividad fotoprotectora y fotorreparadora de los extractos de las angiospermas marinas, Thalassia testudinum Banks ex König (Hyrocharitacea) y Syringodium filiforme Kützing (Cymodoceaceae). Rev. Cienc. Mar. Cos., 8(1), 51-62. http://dx.doi.org/10.15359/revmar.8-1.4
Fagundo-Mollineda, A., Robledo, D., Vásquez-Elizondo, R. M. & Freile-Pelegrín, Y. (2023). Antioxidant activities in holopelagic Sargassum species from the Mexican Caribbean: Temporal changes and intra-thallus variation. Algal Res., 76, 103289. https://doi.org/10.1016/j.algal.2023.103289
Fallarero, A., Peltoketo, A., Loikkanen, J., Tammela, P., Vidal, A. & Vuorela, P. (2006). Effects of the aqueous extract of Bryothamnion triquetrum on chemical hypoxia and glycemia-induced damage in GT1-7 mouse hypothalamic immortalized cells. Phytomedicine, 13(4), 240-245. https://doi.org/10.1016/j.phymed.2003.10.009
Folin, O. & Ciocalteu, V. (1927). On tyrosine and tryptophane determinations in proteins. J. Biol. Chem, 73(2), 627-650. https://doi.org/10.1016/S0021-9258(18)84277-6
González, K. L., Rodríguez, M., Concepción, Á., Valdés, O., Marrero, J. G., Macías-Alonso, M., I., … & Gutiérrez, R. (2017). Phytochemical profile and evaluation of photoprotective potential of Syringodium filiforme kützing. Rev. Cien. Biol. Salud, 19(3), 18-22.
González, K. L., Valdés O., Laguna, A., Díaz M. & González, J. A. (2011). Efecto antioxidante y contenido polifenólico de Syringodium filiforme (Cymodoceaceae). Rev. Biol. Trop., 59 (1), 465-472.
Gupta, A., Singh, R. L. & Raghubir, R. (2002). Antioxidant status during cutaneous wound healing in immunocompromised rats. Mol. Cell. Biochem., 241, 1-7. https://doi.org/10.1023/A:1020804916733
Halliwell, B. (2008). Are polyphenols antioxidants or pro-oxidants? What do we learn from cell culture and in vivo studies?. Arch. Biochem. Biophys., 476(2), 107-112. https://doi.org/10.1016/j.abb.2008.01.028
Hawas, U. W. & Abou El-Kassem, L. T. (2017). Thalassiolin D: a new flavone O-glucoside Sulphate from the seagrass Thalassia hemprichii. Nat. Prod. Res., 31(20), 2369-2374. https://doi.org/10.1080/14786419.2017.1308367
Hernández-Balmaseda, I., Guerra, I. R., Declerck, K., Herrera Isidrón, J. A., Pérez-Novo, C., Van Camp, G., ... & Vanden Berghe, W. (2021). Marine seagrass extract of Thalassia testudinum suppresses colorectal tumor growth, motility and angiogenesis by autophagic stress and immunogenic cell death pathways. Mar. Drugs, 19(2), 52. https://doi.org/10.3390/md19020052
Hoang, H. T., Moon, J. Y. & Lee, Y. C. (2021). Natural antioxidants from plant extracts in skincare cosmetics: Recent applications, challenges and perspectives. Cosmetics, 8(4), 106. https://doi.org/10.3390/cosmetics8040106
Husein el Hadmed, H. & Castillo, R. F. (2016). Cosmeceuticals: peptides, proteins, and growth factors. J. Cosmet. Dermatol., 15(4), 514-519. https://doi.org/10.1111/jocd.12229
IBM Corp. (2020). IBM SPSS Statistics for Windows, Version 27.0. EE. UU.: IBM Corp.
Jafriati, J., Jumadi, O., Hatta, M., Natzir, R., Budu, B., Eddyman, W. F. & Sabir, M. (2019). Analysis of phythology components and potentials of antioxidant activities of Thalassia hemprichii extract. Int. Medical J., 24(3), 145-152.
Jesus, A., Mota, S., Torres, A., Cruz, M. T., Sousa, E., Almeida, I. F. & Cidade, H. (2023). Antioxidants in sunscreens: which and what for?. Antioxidants, 12(1), 138. https://doi.org/10.3390/antiox12010138
Kevrekidou, A., Assimopoulou, A. N., Trachana, V., Stagos, D. & Malea, P. (2024). Antioxidant activity, inhibition of intestinal cancer cell growth and polyphenolic compounds of the seagrass Posidonia oceanica’s extracts from living plants and beach casts. Mar. Drugs, 22(3), 130. https://doi.org/10.3390/md22030130
Lephart, E. D. (2016). Skin aging and oxidative stress: Equol’s anti-aging effects via biochemical and molecular mechanisms. Ageing Res. Rev., 31, 36-54. https://doi.org/10.1016/j.arr.2016.08.001
Lim, H. W., Kohli, I., Ruvolo, E., Kolbe, L. & Hamzavi, I. H. (2022). Impact of visible light on skin health: The role of antioxidants and free radical quenchers in skin protection. J. Am. Acad. Dermatol., 86(3), 27-37. https://doi.org/10.1016/j.jaad.2021.12.024
Lim, S. N., Cheung, P. C. K., Ooi, V. E. C. & Ang, P. O. (2002). Evaluation of antioxidative activity of extracts from a brown seaweed, Sargassum siliquastrum. J. Agric. Food Chem., 50(13), 3862-3866. https://doi.org/10.1021/jf020096b
Lucas, R. M., Yazar, S., Young, A. R., Norval, M., De Gruijl, F. R., Takizawa, Y. & Neale, R. E. (2019). Human health in relation to exposure to solar ultraviolet radiation under changing stratospheric ozone and climate. Photochem. Photobiol. Sci., 18(3), 641- 680. https://doi.org/10.1039/C8PP90060D
Marklund, S. & Marklund, G. (1974). Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem., 47(3), 469-474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Martínez-Daranas, B. & Suárez, A. M. (2018). An overview of Cuban seagrasses. Bull. Mar. Sci., 94(2), 269-282. https://doi.org/10.5343/bms.2017.1014
Mas-Bargues, C., Escriva, C., Dromant, M., Borras, C. & Vina, J. (2021). Lipid peroxidation as measured by chromatographic determination of malondialdehyde. Human plasma reference values in health and disease. Arch. Biochem. Biophys., 709, 108941. https://doi.org/10.1016/j.abb.2021.108941
McKenzie, R. L., Björn, L. O., Bais, A. & Ilyasd, M. (2003). Changes in biologically active ultraviolet radiation reaching the Earth’s surface. Photochem. Photobiol. Sci., 2(1), 5-15. https://doi.org/10.1039/B211155C
Mitchell, C. L. (2016). Oxidative stress and the skin. In D. Armstrong & R. D. Stratton (Eds.), Oxidative Stress and Antioxidant Protection. (pp. 168-182). U.S.A.: John Wiley & Sons, Inc. https://doi.org/10.1002/9781118832431.ch11
Munteanu, I. G. & Apetrei, C. (2021). Analytical methods used in determining antioxidant activity: A review. Int. J. Mol. Sci., 22(7), 3380. https://doi.org/10.3390/ijms22073380
Newman, P. A., Nash, E. R., Kawa, S. R., Montzka, S. A. & Schauffler, S. M. (2006). When will the Antarctic ozone hole recover?. Geophys. Res. Lett., 33(12). https://doi.org/10.1029/2005GL025232
Ohkawa, H., Ohishi, N. & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 95(2), 351-358. https://doi.org/10.1016/0003-2697(79)90738-3
Owen, J. B. & Butterfield, D. A. (2010). Measurement of Oxidized/Reduced Glutathione Ratio. In P. Bross & N. Gregersen (Eds.), Protein Misfolding and Cellular Stress in Disease and Aging: Methods in Molecular Biology (pp. 269-277). U.S.A. Humana Press. https://doi.org/10.1007/978-1-60761-756-3_18
Parker, E. R. (2021). The influence of climate change on skin cancer incidence–a review of the evidence. Int. J. Womens Dermatol., 7(1), 17-27. https://doi.org/10.1016/j.ijwd.2020.07.003
Perry, M. J., Curic, M., Scott, A. L., Ritmejerytė, E., Rahayu, D. U., Keller, P. A., ... & Wangchuk, P. (2024). The In Vitro Antioxidant and Anti-Inflammatory Activities of Selected Australian Seagrasses. Life, 14(6), 710. https://doi.org/10.3390/life14060710
Phandee, S. & Buapet, P. (2018). Photosynthetic and antioxidant responses of the tropical intertidal seagrasses Halophila ovalis and Thalassia hemprichii to moderate and high irradiances. Bot. Mar., 61(3), 247-256. https://doi.org/10.1515/bot-2017-0084
Regalado, E. L., Menendez, R., Valdés, O., Morales, R. A., Laguna, A., Thomas, O. P. & Kijjoa, A. (2012). Phytochemical analysis and antioxidant capacity of BM-21, a bioactive extract rich in polyphenolic metabolites from the seagrass Thalassia testudinum. Nat. Prod. Commun., 7(1). https://doi.org/10.1177/1934578X1200700117
Regalado, E. L., Rodríguez, M., Menéndez, R., Concepción, Á. A., Nogueiras, C., Laguna, A. & Hernandez, Y. (2009). Repair of UVB-damaged skin by the antioxidant sulphated flavone glycoside thalassiolin B isolated from the marine plant Thalassia testudinum Banks ex König. Mar. Biotechnol., 11, 74-80. https://doi.org/10.1007/s10126-008-9123-8
Rivero, F., Fallarero, A., Castañeda, O., Dajas, F., Manta, E., Areces, F. & Vidal, A. (2003). Antioxidant activity in vivo and in vitro of Halimeda incrassata aqueous extracts. Food Sci. Technol., 23, 256-263. https://doi.org/10.1590/S0101-20612003000200026
Sen, C. K. (2003). The general case for redox control of wound repair. Wound Repair Regen, 11(6), 43-438. https://doi.org/10.1046/j.1524-475X.2003.11607.x
Sen, C. K. (2009). Wound healing essentials: let there be oxygen. Wound Repair Regen, 17(1), 1-18. https://doi.org/10.1111/j.1524-475X.2008.00436.x
Svobodová, A., Psotová, J. & Walterová, D. (2003). Natural phenolics in the prevention of UV-induced skin damage. A review. Biomed. Papers., 147(2), 137-145. https://doi.org/10.5507/bp.2003.019
Wuthirak, T., Kongnual, R. & Buapet, P. (2016). Desiccation tolerance and underlying mechanisms for the recovery of the photosynthetic efficiency in the tropical intertidal seagrasses Halophila ovalis and Thalassia hemprichii. Bot. Mar., 59(5), 387-396. https://doi.org/10.1515/bot-2016-0052
Zidorn, C. (2016). Secondary metabolites of seagrasses (Alismatales and Potamogetonales; Alismatidae): Chemical diversity, bioactivity, and ecological function. Phytochemistry, 124, 5-28. https://doi.org/10.1016/j.phytochem.2016.02.004
Zubia, M., Robledo, D. & Freile-Pelegrin, Y. (2007). Antioxidant activities in tropical marine macroalgae from the Yucatan Peninsula, Mexico. J. Appl. Phycol., 19, 449-458. https://doi.org/10.1007/s10811-006-9152-5
1 Department of Marine Resources, CINVESTAV Unidad Mérida, Yucatán CP 97310, Mexico. adrian.fagundo@cinvestav.mx * ORCID: https://orcid.org/0009-0008-3127-5177
2 Institute of Marine Sciences (ICIMAR), Department of Pharmacology. Loma y 37, Nuevo Vedado, Plaza de la Revolución. La Habana, Cuba. ruth.ana30266@gmail.com ORCID: https://orcid.org//0009-0007-5124-758X mariargarcia53@gmail.cu ORCID: https://orcid.org/0009-0003-7301-1157 kethia@icimar.cu ORCID: https://orcid.org/0000-0003-1485-9249 garciateidy@gmail.com ORCID: https://orcid.org/0000-0003-1059-962X ovaldes295@gmail.com ORCID: https://orcid.org/0000-0001-5461-3871 yasnay@icimar.cu ORCID: https://orcid.org/0000-0001-7013-9540 migueldavid@icimar.cu ORCID: https://orcid.org/0000-0003-4175-301X
3 Cuban Neuroscience Center, Calle 190, No 19818, Ave 25 y Ave 27, Playa, La Habana, Cuba. menendezsoto@gmail.com ORCID: https://orcid.org/0000-0002-0889-5174
4 Center for Molecular Immunology. 216 & 15, Atabey, Playa, Cuba. patricial@cim.sld.cu ORCID: https://orcid.org/0000-0001-9164-0238




